Abstract
Plant cellulose and bacterial cellulose has the same chemical structure but different in the mechanical and chemical property. the major problem found in plant-derived is for its highly bound to lignin, forming lignocellulose while bacterial cellulose (BC) is a highly pure and crystalline material generated by different aerobic bacteria, which have very interesting characteristics in comparison to the plant cellulose like being transparent, porous, high water holding capacity, flexible, etc. BC alone or in combination with different components (e.g., biopolymers and nanoparticles) can be used for a wide range of applications in technical textiles such as various medical product, electrical instruments, and food ingredients. Up until now, biomedical devices have gained importance attention due to the increase in medical engineering products for the wound care, regeneration of organs, the diagnosis of diseases and drug transportation. Hence bacterial cellulose has potential applications across several protected areas and permits the development of innovative material. hence overall information about bacterial cellulose synthesis and its applications in medical textile for the various protected areas has been presented here. The latest use of BC in the medical textile field is thoroughly discussed with its application in both a pure and composite form.
Keywords: bacterial cellulose, acetobacterxylinum, gluconacetobacter xylinum, nano-cellulose, wound dressing material.
Introduction
Although the plant is the major contributor of cellulose, various bacteria are able to produce cellulose in a few days as an alternative source. Bacterial cellulose (BC) was produced first by brown (1988) who identified the growth of un-branched pellicle with chemically equivalent structure as plant cellulose. BC structure has only glucose monomer; it exhibits numerous great properties such as unique nanostructure [1]. Bacterial cellulose mainly produced by Acetobacterxylinum. because of its high yield BC production and unique physical and mechanical property, such as high mechanical strength, high crystallinity, high water holding capacity, biodegradable and nanostructure biocompatible polymer [2].Several studies have shown that bacterial cellulose is produced in the form of a pellicle of the desired size and shape on the surface of a culture medium in a static condition, or in the form of gel-like pulp in dynamic condition [3]. The production of bacterial cellulose is receiving great attention for further improvements because of its unique property and wide range application. Therefore researchers have tried to increase the yield of bacterial cellulose from Acetobacterxylinum by using various biochemicals and natural sources [4]. Plant cellulose and bacterial cellulose have the same chemical structure, but different physical and chemical property. Both plant and bacterial cellulose is a linear polymer of d-glucose units linked by β (1→4)-glycoside bonds [5]. Bacterial cellulose (BC) is an environmentally-friendly polymeric material, therefore bacterial cellulose is now receiving more attention in human society. in bacterial cellulose chemical structure, the linear glucan chains form highly regular intra- and intermolecular hydrogen bonds. In the biochemical pathway for synthesis of bacterial cellulose, polymerization and crystallization occur together to include both characteristics. in observation of these Nanofibrils in microscope shown cross-sectional dimensions in the nm range, which can then aggregate to form microfibrils with a width of 50- 80 nm, and a thickness of 3-8 nm[6]. Several studies show that bacterial cellulose (BC) synthesized in abundance by acetobacterxylinum and have vast potential as a novel wound healing system because the high mechanical strength and remarkable physical properties result from the unique nanostructure of the never-dried bc membrane. Bacterial cellulose pellicle (BCP) with AgNp showed significant antibacterial activities of 100% and 99.2% against E.coli and S. Aureus, respectively [7]. Thus BC has wide range applications in various areas such as the textile industry, the paper industry, and the biomedical field as tissue engineering materials due to their interesting property like as good biocompatibility, mechanical properties similar to those of hard and soft tissue and more easy fabrication into a variety of shapes with adjustable interconnecting porosity [8]. The BC pellicle holds 99% water, and this allows room for cell ingrowth and proliferation. If BC is to mimic the collagen fibril network in a blood vessel, it must have a higher strain at break. These changes can be achieved by making the pellicle thicker or dense therefore the potential of bacterial cellulose to function as a scaffold for tissue engineered blood vessels in vitro [9]. Bacterial cellulose is a versatile biomaterial that has the possibility to be modified to receive the desired properties of a scaffold to be used in different tissue engineering applications. One possibility is to modify the BC during the culture time. By varying the culture conditions, BC with different densities in the network of fibrils can be produced [10].BC consists of microfibrils, which are free of lignin and hemicellulose and no need any chemical treatment for purification like plant cellulose. These microfibrils are arranged in a 3d web-shaped structure, providing a porous geometry for high water holding capacity and high mechanical strength. compared to plant cellulose, bacterial cellulose has considerably higher Crystallinity (80–90%),90% water absorption capacity, and degree of polymerization (up to 8000) [11]. These interesting characteristic properties, along with its biocompatibility, make it an attractive candidate for a broad range of applications in various fields, particularly those associated with biomedical and biotechnology applications [12-13].
Material and Method
Organism
Acetobacterxylinum was obtained from the national collection of industrial microorganisms, Pune, India. The culture was maintained on a given medium by NCIM Pune. Agar slants were preserved at 4°c until further sub-culturing.
Preparation of inoculum
The medium for growth and cellulose production consisted of (g/l): sucrose 50, yeast extract 5.0, ammonium sulphate 5.0,monobasic potassium phosphate 3.0, and magnesium sulphate 0.05 [14]. The pH of the medium was adjusted to 5.0 using 1.0 m HCl. The liquid medium (500 ml) in 1-1 conical flask was utilized to grow the initial inoculum for further use. The culture was allowed to grow for a period of 2 weeks at 30°c in a bod incubator. To inoculate every fresh batch of medium, 25 ml of culture was transferred to a volume of 500 ml of standard medium.
Effect of carbon and nitrogen sources on cellulose production
Different carbon and nitrogen sources were used to test their effect on cellulose production. the carbon sources selected for their suitability were sorbitol/ glucose/galactose/lactose/acetic acid/mannitol/maltose/starch or sucrose at a concentration of 50.0 g/l along with 5.0 g/l ammonium sulphate, 5.0 g/l yeast extract, and 0.05 g/l magnesium sulfate. The effect of mannitol and sucrose concentration over the range 70±10 g/l was studied. Furthermore, the following nitrogen sources were used by choosing each of the above carbon sources: casein hydrolysate/ammonium sulphate/glycine/soybean meal/peptone or sodium glutamate at a concentration of 5.0 g/l instead of the ammonium sulphate of the standard medium.
Treatment of cellulose pellicle
The pellicle obtained after 2 weeks of incubation was boiled in 2.0% NaOH solution for 30 min and thoroughly washed with distilled water. Drying was carried out with filter papers and at 60°c in the oven for a period of 6 h. The dry weight of the membrane was measured.
Observation Techniques- BC production by Acetobacter species. The optimal additive was chosen based on the amount of BC produced. The produced BC was analyzed by using X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), thermogravimetric analysis (TGA), Fourier-transform infrared spectroscopy (FTIR), Analysis of Sugars and Inhibitors and dynamic mechanical analysis (DMA)[15-18].
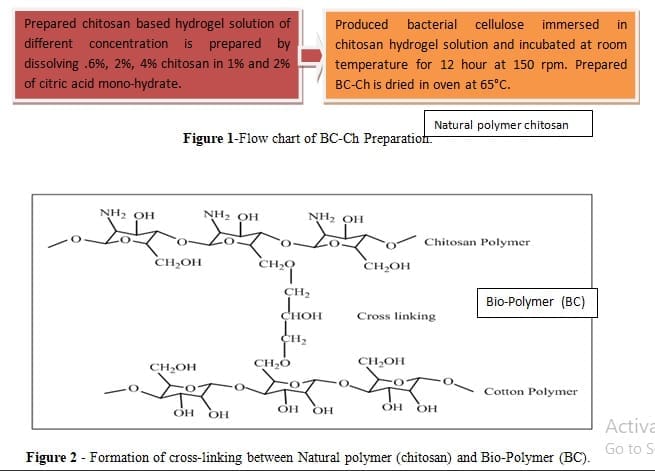
Application in Medical Textiles
Bacterial cellulose is a biopolymer with very interesting properties such as high water holding ability, high crystallinity, high purity, high efficiency of antimicrobial activity to plants cellulose. Because of these properties, We have developed BC based antimicrobial bandage.
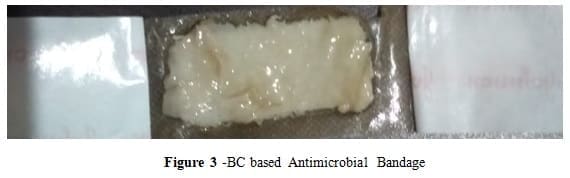
After review the several reviews of several research papers, I have observed that BC has a wide range of biomedical applications because of its very interesting properties. BC is an excellent wound dressing device, eliminating exudates, avoiding infections and reducing local pain. Several studies show that wound dressing material is basically used for avoiding infection, which is caused by high bacterial levels. So a new strategy is used for developing BC-Ch Based wound dressing with high antimicrobial efficiency. Chitosan-based Bacterial cellulose has high antimicrobial Efficiency to Chitosan-based plant cellulose and low irritation when wrap on burn area or cut mark of the body [19-20]. So we can develop a new product (antimicrobial bandage) which will be used in a small injured area of a human’s body. The crystallinity of bacterial cellulose makes more durable in the face of chemical reactions and the mechanical pressure one might find in the body. The wound dressing has been on the market for more than a year and already has been used on thousands of patients. When the bacteria are grown properly, the cellulose has the unique ability to moisten dry areas, while at the same time absorbing unwanted fluids leaking from the wound. This helps create an optimal environment for wound healing. The dressing’s capability to manage moisture promotes the recovery of normally difficult-to-heal ulcers and reduces pain.
Conclusions
Modified Chitosan-based bacterial cellulose has good Antimicrobial activity efficiency against E.coli and S.aureus to the modified Chitosan-based plant cellulose. Several studies show that bacterial cellulose has very interesting properties; Hence Bacterial cellulose has potential applications in the biomedical field across several Protective areas like as wound dressing, drug delivery, artificial skin vascular prosthesis and tissue engineering scaffold [21]. Other cellulosic material production process creates a great amount of pollution like as the paper and cellulose industry that’s why the new technology is investigated by the researcher for producing environment-friendly cellulose. Bacterial cellulose has potential application in tissue engineering because it’s in situ moldability, good biocompatibility, high water holding capacity, and high resistance to in-plane tensile deformation and Inherent limitations are its resistance to biodegradation, low resistance to perpendicular compressive deformation, and low porosity. The highlighted studies demonstrate successful strategies to exploit the in situ moldability, overcome the resistance to biodegradation, improve mechanical properties, increase porosity, and impart bioactivity, paving the way toward BC-based scaffolds with improved functionality and tipping the balance of merits and limitations in [22]. Our results have shown that wounds are very well isolated from the outside environment by application of the MC membranes. Due to its unique 3D nanostructure, the MC membrane can virtually replicate the wound surface at the Nanoscale level and create optimal moist conditions for wound healing and skin regeneration. During the clinical trials, MC dressings were very well tolerated by patients, significantly reducing pain during treatment and allowing for painless removal of the dressing from the wound. In shallow wounds, MC dressing promoted the growth of the epidermis and in deep wounds shortened the period of scab demarcation. In our opinion, treatment with MC dressings should be continued until an entirely new epidermis appears; otherwise, second necrosis can take place. The MC dressing needs to be changed every day or rewetted to maintain the desired moist environment. Drying of the dressing on the wound causes the effect of squeezing (pressing). The results to date suggest that MC dressing might be effective in reducing scar formation. In our opinion, cellulose dressings should be applied to fresh wounds immediately after burn injury. Considering this and the capability to produce MC dressing of different sizes and shapes, we think that, in addition to hospitals and ambulatories, MC should be widely used in all emergency responding units such as police, emergency, firemen, army, etc. The unique features of the MC have been demonstrated to be effective in the burn wound healing response [23]. The research on BC itself as well as its derivatives are directed to special applications in the field of biomaterials. At present, our efforts are concentrated on possibilities for the utilization of chemically unmodified BC. Among other uses, cellulose produced by Acetobacterxylinum is applicable in some medical areas, for instance as a temporary skin substitute in the treatment of skin wounds, as a surgical wipe, treatment pad, burn bandage or tissue/organ drape[24].
Future perspective
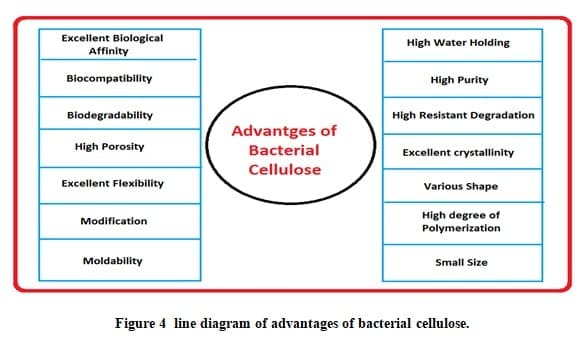
I have observed after review several research papers that BC has wide range application in protective textile. Bacterial cellulose has a higher water holding ability, higher strength, high purity, not easily burn and environment-friendly to the plant cellulose. So it can be used as liner fabric in place of cotton fabric in firefighter suit and other armor fabric. Bacterial cellulose can be used for commercialization purpose but the main drawback of bacterial cellulose is yield (g/l). So many researchers are trying to improve the yield of bacterial cellulose. Now the highest yield of bacterial cellulose is 15 g/l by 1.4% of ethanol in culture media. There are three advantages of bacterial cellulose over the plant cellulose, first minimum time to produce, high crystallinity of BC and its 3 D porous structure with 69-90% water holding capacity. Plant cellulose and bacterial cellulose have the same chemical structure, but different physical and chemical property. Bacterial cellulose has several advantages over the plant cellulose that’s given below.
References-
[1] Klemm, D., Schumann, D., Udhardt, U. and Marsch, S., (2001). Bacterial synthesized cellulose—artificial blood vessels for microsurgery. Progress in Polymer Science, 26(9), pp.1561-1603
[2] Stumpf, T.R., Yang, X., Zhang, J. and Cao, X., (2018). In situ and ex situ modifications of bacterial cellulose for applications in tissue engineering. Materials Science and Engineering: C, 82, pp.372-383.
[3] Liu, M., Li, S., Xie, Y., Jia, S., Hou, Y., Zou, Y. and Zhong, C., (2018). Enhanced bacterial cellulose production by Gluconacetobacter xylinus via expression of Vitreoscilla hemoglobin and oxygen tension regulation. Applied microbiology and biotechnology, 102(3), pp.1155-1165.
[4] Gullo, M., La China, S., Falcone, P.M. and Giudici, P., (2018). Biotechnological production of cellulose by acetic acid bacteria: current state and perspectives. Applied microbiology and biotechnology, 102(16), pp.6885-6898.
[5] Chen, S.Q., Lopez-Sanchez, P., Wang, D., Mikkelsen, D. and Gidley, M.J., (2018). Mechanical properties of bacterial cellulose synthesised by diverse strains of the genus Komagataeibacter. Food hydrocolloids, 81, pp.87-95.
[6] Shi, Z., Zhang, Y., Phillips, G.O. and Yang, G., (2014). Utilization of bacterial cellulose in food. Food hydrocolloids, 35, pp.539-545.
[7] Wu, C.N., Fuh, S.C., Lin, S.P., Lin, Y.Y., Chen, H.Y., Liu, J.M. and Cheng, K.C., (2018). TEMPO-oxidized bacterial cellulose pellicle with silver nanoparticles for wound dressing. Biomacromolecules, 19(2), pp.544-554.
[8] Torgbo, S. and Sukyai, P., (2018). Bacterial cellulose-based scaffold materials for bone tissue engineering. Applied Materials Today, 11, pp.34-49.
[9] Kwak, M.H., Kim, J.E., Go, J., Koh, E.K., Song, S.H., Son, H.J., Kim, H.S., Yun, Y.H., Jung, Y.J. and Hwang, D.Y., (2015). Bacterial cellulose membrane produced by Acetobacter sp. A10 for burn wound dressing applications. Carbohydrate polymers, 122, pp.387-398.
[10] Helenius, G., Bäckdahl, H., Bodin, A., Nannmark, U., Gatenholm, P. and Risberg, B., (2006). In vivo biocompatibility of bacterial cellulose. Journal of Biomedical Materials Research Part A: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials, 76(2), pp.431-438.
[11] Hallac, B.B. and Ragauskas, A.J., (2011). Analyzing cellulose degree of polymerization and its relevancy to cellulosic ethanol. Biofuels, Bioproducts and Biorefining, 5(2), pp.215-225.
[12] Hussain, Z., Sajjad, W., Khan, T. and Wahid, F., (2019). Production of bacterial cellulose from industrial wastes: a review. Cellulose, 26(5), pp.2895-2911.
[13] Joseph, G., Rowe, G.E., Margaritis, A. and Wan, W., (2003). Effects of polyacrylamide‐co‐acrylic acid on cellulose production by Acetobacterxylinum. Journal of Chemical Technology & Biotechnology: International Research in Process, Environmental & Clean Technology, 78(9), pp.964-970.
[14] Watanabe, K. and Yamanaka, S., (1995). Effects of oxygen tension in the gaseous phase on production and physical properties of bacterial cellulose formed under static culture conditions. Bioscience, biotechnology, and biochemistry, 59(1), pp.65-68.
[15] Cheng, K.C., Catchmark, J.M. and Demirci, A., (2009). Effect of different additives on bacterial cellulose production by Acetobacterxylinum and analysis of material property. Cellulose, 16(6), p.1033.
[16] Kiziltas, E.E., Kiziltas, A. and Gardner, D.J., (2015). Synthesis of bacterial cellulose using hot water extracted wood sugars. Carbohydrate polymers, 124, pp.131-138.
[17] Molina-Ramírez, C., Castro, M., Osorio, M., Torres-Taborda, M., Gómez, B., Zuluaga, R., Gómez, C., Gañán, P., Rojas, O. and Castro, C., (2017). Effect of different carbon sources on bacterial nanocellulose production and structure using the low pH resistant strain Komagataeibactermedellinensis. Materials, 10(6), p.639.
[18] Kamarudin, N.S.B., Rahman, N.A., Kalil, S.M. and Kamarudin, S.K., (2018). Comparative Study of Bio-Cellulose from AcetobacterXylinum 0416 and Commercial Hard Gelatine Capsule. International Journal of Applied Engineering Research, 13(1), pp.743-748.
[19] Mohite, B.V., Koli, S.H. and Patil, S.V., (2019). Bacterial Cellulose-Based Hydrogels: Synthesis, Properties, and Applications. Cellulose-Based Superabsorbent Hydrogels, pp.1255-1276.
[20] Moniri, M., BoroumandMoghaddam, A., Azizi, S., Abdul Rahim, R., Bin Ariff, A., ZuhainisSaad, W., Navaderi, M. and Mohamad, R., (2017). Production and status of bacterial cellulose in biomedical engineering. Nanomaterials, 7(9), p.257.
[21] Lin, W.C., Lien, C.C., Yeh, H.J., Yu, C.M. and Hsu, S.H., (2013). Bacterial cellulose and bacterial cellulose–chitosan membranes for wound dressing applications. Carbohydrate polymers, 94(1), pp.603-611.
[22] Gatenholm, P. and Klemm, D., (2010). Bacterial nanocellulose as a renewable material for biomedical applications. MRS bulletin, 35(3), pp.208-213.
[23] Czaja, W., Krystynowicz, A., Kawecki, M., Wysota, K., Sakiel, S., Wróblewski, P., Glik, J., Nowak, M. and Bielecki, S., (2007). Biomedical applications of microbial cellulose in burn wound recovery. In Cellulose: Molecular and structural biology (pp. 307-321). Springer, Dordrecht.
[24] Roman, M., Haring, A.P. and Bertucio, T.J., (2019). The growing merits and dwindling limitations of bacterial cellulose-based tissue engineering scaffolds. Current Opinion in Chemical Engineering, 24, pp.98-106.
AshutoshPandey, Annika Singh, Mukesh Kr. Singh, Supriyo Chakraborty
Uttar Pradesh Textile Technology Institute Kanpur India
Institute of Biosciences and Biotechnology, CSJMU Kanpur

